Health-&-Fitness
Alzheimer's Disease: Symptoms, Causes, Preventions
Thor actor Chris Hemsworth has decided to take a break from acting after discovering the risk of Alzheimer's. This news has shocked millions of fans worldwide. Now, what is Alzheimer's? It is a neurological ailment that causes cognitive decline and memory loss. Alzheimer's disease is characterized by the death of brain cells. If you are experiencing frequent memory loss, this should not be ignored. Let’s take a look at the symptoms, causes, and preventive measures of Alzheimer's disease.
What is Alzheimer's?
Alzheimer's disease is an incurable and irreversible brain condition. The condition has a profound impact on a person's memory, reasoning, learning, and organizational abilities, and ultimately impairs their capacity to do routine everyday tasks. Alzheimer's disease is not a typical component of aging.
The symptoms of Alzheimer's disease worsen with time. In fact, experts estimate that the disease process may continue for at least ten years before the onset of Alzheimer's clinical manifestations.
Read More: Safe Toys for Children: Tips to avoid harmful playing objects
German psychiatrist Alois Alzheimer, a pioneer in correlating symptoms to microscopic brain changes, tells the eerie case of Auguste D., a woman with severe memory loss, unjustified suspicions about her family, and other deteriorating psychological abnormalities.
At her autopsy, he saw significant shrinkage and aberrant deposits surrounding nerve cells in her brain. Dr. Alzheimer passed away in 1915, never imagining that his experience with Auguste D. would impact the lives of millions and spark a large worldwide research initiative.
Dr. Alzheimer is revered by scientists not just for his ground-breaking description of a serious illness, but also as a role model. He created a new benchmark for comprehending neurodegenerative illnesses by developing a deep therapeutic interaction with his patients and using innovative scientific methods to assess how symptoms connected to physical brain changes manifest.
Read More: 7 Homemade Drinks for Sore Throat
Symptoms and Signs of Alzheimer's
Alzheimer's disease is characterized by memory loss. Recent talks and occurrences may be difficult to recall at an early stage. As the illness advances, memory deficits increase, and more symptoms emerge.
Initially, a person with Alzheimer's disease may be aware of memory and thinking organization difficulties. A family member or acquaintance may be more likely to observe the worsening of symptoms.
Changes in the brain caused by Alzheimer's disease exacerbate problems with:
Memory
Everyone has periodic memory lapses, but Alzheimer's disease is characterized by persistent and worsening memory loss that impairs the capacity to perform at work or at home.
Read More: Food, Drinks during Dengue: What to consume, what to avoid
Individuals with Alzheimer's may repeat comments and inquiries repeatedly. Forget talks, appointments, and activities, and subsequently do not recall them. Frequently misplace belongings, frequently placing them in odd places. Get lost in familiar surroundings. Eventually, forget the names of loved ones and commonplace items.
Multitasking
Managing funds, balancing checkbooks, and paying bills on time may be very demanding, and multitasking is especially difficult. A person with Alzheimer's may eventually lose the ability to identify and manipulate numbers.
Making choices and judgments
Alzheimer's disease diminishes the capacity to make appropriate choices and judgments in daily circumstances. A person may, for instance, make bad or unusual decisions in social encounters or wear unsuitable clothing for the weather. It may be more challenging to react successfully to routine concerns, such as food on the stove that is burning or unexpected driving scenarios.
Read More: Conjunctivitis: Causes, Symptoms, Treatment and Prevention
Planning and completing routine duties
As the illness develops, tasks that formerly required sequential steps, such as planning and making a meal or playing a beloved game, become difficult. People with severe Alzheimer's disease often forget how to do fundamental functions such as dressing and washing.
Alterations in personality and conduct
Moods and behaviors may be affected by Alzheimer's disease-related brain alterations. Because these abilities are governed by regions of the brain impacted later in the progression of the illness, they may be retained for longer.
Causes of Alzheimer's Disease
Alzheimer's disease is caused by aberrant protein accumulation in the brain. The accumulation of these proteins, which are known as amyloid protein and tau protein, causes cell death.
Read More: Mental Health: Types of Mental Illness and supporting someone with a mental health problem
In addition to nerve cells, the human brain includes around 100 billion different types of cells. The nerve cells collaborate to carry out all the communications required for cognitive processes such as thinking, learning, remembering, and planning. Scientists think that amyloid protein accumulates in brain cells to create plaques, which are bigger aggregates.
Tau forms tangles composed of twisted fibers of another protein. These plaques and tangles impede the connection between nerve cells, preventing them from performing their functions. Alzheimer's disease symptoms are caused by the gradual and progressive loss of nerve cells, which begins in one region of the brain and spreads to others.
How to Minimize the Risk of Alzheimer's
There are no proven preventative methods. At present, the most effective means of preventing cognitive decline are behaviors that promote health.
Read More: Protecting Your Child’s Mental Health: 10 Tips for Parents
The following steps may be useful:
Attempt to stop smoking:
If you are a smoker, quitting has both immediate and long-term health advantages.
Exercise frequently:
Physical activity decreases the risk of several diseases, including cardiovascular disease and diabetes.
Keep your mind engaged:
Try some mental conditioning activities.
Eat healthily:
Consume a properly balanced diet with the right amount of fruits and vegetables.
Read More: World Mental Health Day: What effect does depression have on the performance of athletes?
Maintain a socially engaged lifestyle:
It is probable that friendships, volunteering, and hobbies will help your general health.
Before making significant changes to your lifestyle, consult with your physician.
Treatment of Alzheimer's
Although there is no cure, several therapies might temporarily alleviate symptoms. Many individuals with Alzheimer's disease learn to manage their symptoms and enhance their quality of life.
Medications
Cholinergic medications may temporarily alleviate mild to severe Alzheimer's disease's cognitive symptoms. These function by raising the concentration of acetylcholine, a brain neurotransmitter that helps reestablish communication between brain cells.
Other drugs alleviate behavioral problems including insomnia, agitation, anxiety, and sadness. Although these therapies do not directly cure Alzheimer's disease, they may enhance the quality of life.
Read More: Spending Money for Happiness: 10 Effective Ways
Lifestyle changes
A person with Alzheimer's disease should, to the extent feasible, maintain normal social contact with friends and family, engage in regular exercise, and participate in activities that stimulate the brain. Consult a physician if you have any safety-related concerns (such as driving abilities).
Verdict
The disorder known as Alzheimer's disease is one that causes neurodegeneration. Memory loss and cognitive decline are caused by the accumulation of plaques and tangles in the brain, in addition to the death of brain cells.
At this time, there is no known therapy that will reverse the condition, but there are medications and other therapies that may assist improve a person's cognitive, psychological, and behavioral symptoms, as well as their quality of life.
Read More: National Mental Health Strategy 2020-2030: Towards ensuring quality mental healthcare
So far, we have discussed the signs, reasons, and prevention and treatment of Alzheimer's disease. Hope it helps!
10 Rooftop Gardening Tips for this Winter
A rooftop garden is something more than a collection of plants. A garden offers a place of serenity on the rooftop of a flat or apartment building. Especially, under the winter sun, people of all ages enjoy spending time in a roof garden. With the proper planning, you can not only grow plants in your roof garden, but also make the space suitable for social gatherings, romantic evenings, and family time. Let’s take a look at some effective tips for winter roof gardening.
10 Effective Tips to Start a Roof Garden
Urban rooftop gardens are an excellent idea, especially in winter. But before you start planting, you need to consider several things in winter rooftop gardening. Check here for the best and perfect tips to get started.
Building Code
First, check your building codes to ensure that your local building codes will allow you to plant on the roof. If you are living in a rented apartment, talk to the building owners to see whether they allow gardening in the shared space.
Read More: IFAD supports smallholder farmers with nutritious homestead vegetable gardening kits
Access
Do you even think about access to your materials and supplies in and out? If you live in an apartment or flat and on a higher floor like 6th or 7th or more than the higher floor, make sure you can use the elevator.
Privacy
Many rooftops are adjoined by neighboring buildings. If your rooftop garden can disturb others, it won’t be honest at all. So make sure your garden is just in your area.
Containers
Containers are one of the most artistic items in a rooftop garden. Take time and choose and select your rooftop containers patiently. Besides, you need to keep in mind the size, weight, and material when choosing your rooftop containers.
Read More: Possible Food Crisis: Vegetable gardening at courtyards of 6.5 lakh houses planned
Remember, you will need containers that are large enough for the roots of whatever plants you choose. As the amount of soil needed will vary, research your choices before you plant them. If you search, you will find a lot of plants that will be labeled as “Great for containers.”
The weight of the container is a great issue as you need to think about how much your rooftop can support. Containers become heavier when you water them.
Containers made with traditional materials; clay, terra cotta, and cement are quite heavy. However, plastic pots and the newer synthetic containers are light naturally enough. You can lift them easily. Make sure you also consider balancing the height of your plants. For a windy rooftop, you have to choose heavier containers.
Read More: 10 houseplants that are bad for your pets
Soil
Some think about soil in the last when they are planning to make a garden. But soil should be the first and most important part of your garden. Remember, you need good soil for healthy plants. When your soil is good and fertile, you don’t work hard for your garden.
If you are planning to grow your plants in containers and raised beds, you will have the advantage of bringing in soil. You will have huge good potting mixes on the market. Besides, you can combine 3 parts of compost to make good manure. Make sure you make a hole in each pot so that additional water runs away and keep the soil moderately wet as so much wet soil makes the plants spoil.
Plants
When you are going to choose a rooftop plant, you should consider some things. So, you have to choose drought-tolerant plants for rooftop gardens as most rooftops get very hot during the day. Besides, don’t forget to consider the sun exposure and hardiness zone.
Read More: Top 10 Bathroom Plants to grow in humid environment of shower
Rooftops hold heat and so they create weather that drifts about a zone higher than a garden on the ground would be. Therefore, you have to plant plants that can handle the heat.
As a list of certified rooftop plants is not available, you need to follow some tips to choose rooftop plants. You can avoid tall trees and focus on small trees for your winter roof gardening.
You can choose plants with limited root systems that don’t need a lot of soil. They can be herbs, vegetables, and smaller to mid-sized perennials. Remember trees and shrubs require more soil, larger pots, and above all more caring that you may not afford in your busy life.
Read More: How Can One Person Reduce Environmental Pollution?
Fertilizer
You must remember that Container plants will need regular fertilizer. A great potting mix can be exhausted over time, as plants take up the nutrients and the water leaches them out. When a plant grows and becomes large, it requires more water. The type of fertilizer you use will direct you on how often and how much you will need to fertilize through its instructions. However, fertilizing every 2–3 weeks is sufficient.
You will find many good fertilizers on the market. Make sure you choose more organic ones. A water-soluble fertilizer is also available in the market and you can choose it as it is the fastest way to get the nutrients to your plants.
Tools
You don’t need so many tools for a rooftop container garden as you would for a traditional ground-level garden. As you have to do a lot of scooping and filling, you need just a trowel and perhaps a soil scoop at first. Moreover, a small tarp will be essential for you when you are emptying soil, to keep from making a mess.
Read More: Healthy plants can protect environment, boost economies: FAO
Plus, the other fundamental tool you need is a good pair of pruners, to prune and clean plants. Actually, the tools you require depend on what you’re growing and how much maintenance you intend to do.
However, you have to also remember that a rooftop garden will be subject to pests and problems also. As insects can fly and can spore, you will need to monitor your plants and try to catch problems while they’re small. You can keep your plants healthy and stress-free by feeding and watering them well. By maintaining all the possible ways you will limit their problems.
Electrical Wiring
Electricity isn’t mandatory but if you have an electrical connection to your roof, it will be easier for you to take care of your plants at night and also when you are busy in the daytime. Besides, you can enjoy the beauty of your garden with your family or lovely ones. However, if it is not possible to electrify your rooftop, you can use candles that are the best lighting for weeding and enjoying the beauty of the garden.
Read More: Grow more food, keep enough stock to avert any crisis: PM Hasina
Make a Plan
Check the space size of your rooftop as you have to put a lot of things associated with gardening including tools, fertilizer, compost, buckets, etc. If the space of your rooftop is limited, it will be hard for you to keep all things in the limited area. When your space is limited, you can consider the shelves that will help you to cover the area.
However, you have to account for how much you are willing to spend. Consider your budget first and then make a plan for your winter roof gardening. Remember, you need to buy some essential things to make a roof garden. Plus, you may need to do more structural work to support and take care of your gardening to make it look elegant.
Bottom Line
It is a great idea to make a roof garden in winter but make sure you do proper research to learn how to cultivate fresh herbs and vegetables, keep the plants alive, use compost to make your plants healthy, and use insecticides to free your garden from pests. Seasonal roof terrace design ideas can also help you to build an amazing garden this winter.
Read More Ban on cutting trees in reserved forests extended until 2030
How to Clean Baby Bottles
Many parents choose bottle feeding for their babies. If you don’t compromise your child’s health and safety, it's requisite and important to properly wash and sanitize the baby’s feeding equipment. The process includes cleaning or sterilizing each bottle with the teat, and screw cap after every feed until they are at least 12 months old. Let’s take a look at the proper tips and tricks to n and sterilize baby bottles.
Health risks of babies for using unsanitized feeding bottles
Babies under 3 months old are more vulnerable to viruses and bacteria as their immune systems are underdeveloped. The South Australian Women's and Children's Health Network stresses that it is important to sterilize bottles to prevent sickness in infants up to the age of 6 months — or even up to a year. If the bottle is not washed or poorly washed bottles after each feed, it may expose your baby to certain risks, including:
Tooth Decay
Generally, tooth decay in babies can occur as the result of vulnerability to liquids for a long time like if you allow them to sleep with a bottle that contains juice. When a baby sleeps with keeping juice or liquid like milk in their mouth, this liquid allows acid-producing bacteria to build up in their mouth and cause teeth decay.
Read More: Safe Toys for Children: Tips to avoid harmful playing objects
According to the Northern Ireland Public Health Agency, formula milk contains bacteria and they can multiply if a bottle is left at room temperature.
Rotavirus
Rotavirus is a type of stomach bug that can spread from touching contaminated objects or surfaces. So, it is imperative for you to wash your hands frequently, especially before preparing baby bottles.
Cronobacter Infections
According to the Centers for Disease Control and Prevention (CDC), the national public health agency of the USA, Cronobacter germs can contaminate dry, powdered foods including infant formula and, subsequently, baby bottles.
Read More: Top 10 Most Expensive Foods in the World
This type of bacteria can cause a very rare but life-threatening blood infection called sepsis. Sepsis can happen particularly in infants.
Right ways to keep baby bottles & other feeding equipment clean
Properly Wash Your Own Hands
Many people forget this important and crucial first step. Research has shown that about 97% of people don’t wash their hands properly before preparing baby bottles. For this reason, most of the cross-contamination happens. Therefore, make sure you have thoroughly sanitized your hands before preparing baby bottles.
However, if you are about to feed the baby with a bottle while staying outside the home, try to use sanitizer or find the nearest restroom to wash your own hands thoroughly with soapy water so that you can prevent yourself from transmitting germs onto the bottle or nipple.
Read More: 12 foods that shouldn’t be given to babies and children
Wash Bottle Parts Separately with Warm Water
You need to wash every and each part of the baby bottles. Don’t focus on the type of bottle you're using. Separate every component and wash each part properly.
Then, rinse each part of your bottle under running Water. Though you can rinse the baby’s feeding equipment under cold or warm water. It is wise to rinse the bottles and equipment under warm water. Warm water helps to kill germs.
Scrub in Soapy Water
When your rinsing the bottle parts under running water is done, fill a separate basin with hot water and soap. You need to use a dedicated brush to scrub each part individually. However, make sure you use this brush to clean the baby bottles only.
Read More: How to entertain an unwell child without screen time
You can also use a good dishwasher instead of soap to wash every part of the baby’s feeding equipment.
You can dip the bottle parts under the dishwasher mixed water or soapy water for some time.
When brushing is done, rinse each part of the bottles and other feeding tools under running water again.
Dry on a Rack or Clean Space
After washing dry every part of the bottle or feeding equipment with a clean paper towel or dish towel, and store them in a dust-free, clean space. Let them air dry but not patting them dry, as patting them dry can transfer germs from the towel to the newly cleaned bottle.
Read More: Best foods for expecting mothers
Other Ways of Sanitizing Baby Bottles
There are several ways you can sterilize, sanitize and clean your baby bottle:
Boiling sterilization
Boiling is the most reliable way of sterilizing baby bottle-feeding equipment. So boil all equipment after every use or within 24 hours of use.
First, take the washed bottles, teats, rings, and caps in a large pot and fill the pot with sufficient water so that everything is covered. Make sure all air bubbles are disappeared. Then boil everything for 5-6 minutes. When everything is cool, take them out with tongs or clean hands. Throw off excess water. It is not necessary to dry the bottles. Finally, store them in a clean container in the fridge.
Read More: How to deal with your demanding child?
Chemical sterilization
This is the way where you can sterilize baby bottles with an antibacterial solution that is available in liquid or tablet form. This is one type of bleach that is mixed easily with water. Although it is enough strong to kill bacteria, it’s safe for your baby.
First, you must follow the manufacturer’s instructions carefully to make up the solution the right way. Remember, the solution will work correctly when it’s made at the right strength. Completely drown-washed bottles, especially every part of the bottles including teats, rings, and caps.
Let everything keep in the solution for the recommended time. You can keep the equipment in the solution for the whole day. Remove equipment using tongs. Remove excess solution, but don’t need to dry the equipment. Store everything in a clean container and put the container in the fridge.
Read Top brain foods for studying and exams
Steam sterilization
In this system, you heat your equipment to a temperature high enough to kill bacteria. First, keep clean equipment in the unit and add water following the manufacturer’s instructions. Then, on the switch.
As it is automated, the unit switches itself off when the work is completed. Store equipment in a clean container and you can follow the manufacturer’s instructions to know the exact time you can store sterilized equipment.
Microwave sterilization
Microwave sterilization is like steam sterilization.
First, see the instructions carefully and check the microwave power needed. However, make sure you don’t put anything made of metal inside these sterilizers.
Read More: How physical punishment affects children and alternative ways to discipline them
Don't put your equipment straight into the microwave to sterilize it because this won’t work rather your equipment will melt or get damaged.
Bottom Line
So far we have discussed the simple steps to clean baby bottles efficiently. You can also follow the sterilization methods to get your baby's bottle and other feeding types of equipment sanitized and ready for use to avoid the risks of bacterial infection or contamination. Don’t forget to clean the bottle just after every feeding to prevent any bacterial contamination.
Safe Toys for Children: Tips to avoid harmful playing objects
Kids are always happy with their toys. The right and safe toys can provide your child with so many benefits like encouragement to play, stimulation of their senses, support in developing skills, and more. However, some toys are harmful to kids. Let’s take a look at how to choose safe toys for children.
How Toys Can Harm Babies
Toys can harm babies in many ways. For instance, -Babies may feel choked while playing with balloons or toys containing strings and wires. -The heavy large toys can fall upon small kids-Electric toys can give them shocks. -The sharp-edged toys can cause injury to children-Babies can swallow small playing objects like magnets, marbles, jewelry, etc. -Dirty toys can affect the kids with bacteria. -Noisy toys can scare young kids.-Babies may hurt themselves or other kids with shooting toys.-Toys built with harmful materials can cause harm to kids’ health.
Read More: 12 foods that shouldn’t be given to babies and children
While playing with toys, a child may experience serious injury or even death especially when the toy doesn’t suit the child’s age or includes dangerous elements, or is improperly built or used in the wrong way. Although, toy-injuries are usually minor cuts, scrapes, and bruises parents should be careful.
13 Guidelines to Choose Safe Toys for Babies
Age-Appropriate Toys
Generally, toys are designed according to the age of the baby. Age is mentioned on the cover of the toy. So, keep in mind the age of your baby whenever you wish to buy a toy. Age-appropriate toys are safe for your child and help in the better development of your child.
Size-Appropriate Toys
Size is one of the major points you need to consider while buying a toy for your child. Make sure you select the size of the toy according to your child’s age, height, size, nature, and health status. Naturally, babies love to keep their favourite toys close to their bodies, especially their noses and mouths.
Read More: How to entertain an unwell child without screen time
Parents should avoid choosing too small toys for young babies as such toys can cause choking when taken in the mouth. Small objects like flashing decorations or jewelry can be harmful to young children.
On the other hand, too large and heavy toys for your child’s size can be difficult for him or her to handle. There is a risk that during playing the toy may fall on the child and hurt him/her.
Chewing Resistant
Naturally, children have the curiosity to put everything in their mouths. This habit can cause bad accidents that can be life-threatening too. So, you have to choose chewing-resistant toys for your child so that they won’t swallow them.
Read More: How to deal with your demanding child?
Washable Toys
Children are not careful about their toys and so they throw their toys here and there while playing. When a kid throw his/her toy in a dirty place, and pick it up again, the toy may carry several bacteria.
Those bacteria may cause bacterial infections as the kids have the habit of putting their toys in their mouths. So, it’s wise to choose toys that are easily washable and you can sanitize from time to time.
Avoid Loud Toys
The main function of toys is to entertain your child. But too loud or screaming toys can hardly give pleasure to your child rather they can irritate them by causing noise pollution. Most importantly, they can harm the listening power of your child. So, don’t buy toys that are too loud for your little one.
Read More: How physical punishment affects children and alternative ways to discipline them
No Strings Or Wires
Don’t buy toys that have long strings, attached ribbons, or frills. If you have found some, remove such kind of material immediately from those toys as your child may wrap it around him/her neck and feel choked. Besides, it may harm the child’s blood circulation by restricting the flow of the blood through his/her veins.
Photographing the birth of a nation and its architect
Renowned French photographer Anne de Henning, who documented Bangladesh during its birth and infancy, said she found Bangabandhu Sheikh Mujibur Rahman to be “very charismatic”.
Talking about her photo of Bangabandhu’s speech at the first council of Awami League in independent Bangladesh, she said it was a strong testimony to the victory of freedom over oppression.
An exhibition titled ‘Witnessing History in the Making: Photographs by Anne de Henning’ is being held at Guimet Museum of Asian Art in Paris. The exhibition has been produced by Samdani Art Foundation and Centre for Research and Information (CRI).
Radwan Mujib Siddiq, grandson of Father of the Nation Bangabandhu Sheikh Mujibur Rahman and trustee of CRI made a visit to the exhibition.
He said, “There was a leader from the subcontinent who led his country to independence based on very progressive ideals. He wanted a country that was secular and equal for all. And the whole of Bangladesh got behind him, fought a war against all odds and Bangladesh emerged independent.”
Radwan said Bangabandhu had a connection with the people and he could represent them the way that politicians are supposed to.
Read more: Radwan Mujib visits Paris photo exhibit on Bangladesh’s birth, Bangabandhu
Henning said she was shocked to learn that three years after she photographed Bangabandhu, he was assassinated with most of his family members.
The exhibition is being held from October 19, 2022 to January 23, 2023 in partnership with Asia Now Paris Art Fair and Guimet Museum of Asian Art, according to a press release.
“The whole art world is in Paris right now and we're so honoured that this exhibition has been invited by the Guimet Museum,” said Nadia Samdani MBE, President and Co-founder of Samdani Art Foundation
Reminiscing the days of Bangladesh’s Liberation War, Anne De Henning said, “You just had to walk through the streets and you would see the people gearing up, poorly equipped, but they expressed such a will and determination of achieving freedom for their country that it was extremely moving.”
Henning said the images at the exhibition should inspire the feeling that courage and determination can get you a long way.
The first iteration of the exhibition was held in Dhaka from December 10, 2021 to March 31, 2022 to celebrate 50 years of Bangladesh’s independence.
Henning’s photographs from 1972 feature Bangabandhu Sheikh Mujibur Rahman. She captured Bangabandhu giving a speech at the first Council Meeting of Awami League after the independence of Bangladesh. “I came specifically from Calcutta to photograph the event,” she said. Although at the time she favoured shooting in black and white, she chose to capture this event in colour because of the vibrant blue, white and red stripes of the shamiyana — ceremonial tent — that housed the event.
7 Homemade Drinks for Sore Throat
As winter is approaching, many people are experiencing conditions like sore throat and scratchy throat. It causes constant itchy feelings, irritation, and difficulty in swallowing. Don’t worry! Some drinks can relieve the scratchy, and painful feelings. Let’s take a look at home remedies that kills a sore throat fast.
7 Healthy Drinks to Treat Sore Throat
Warm honey-lemon water
Warm water can help settle a sore throat. Hot water is special to soothe a sore throat and can help provide relief for cough, and runny nose.
However, warm water with lemon may be especially useful as it can add a small extra dose of vitamin C to pacify your sore throat. Vitamin C is great to boost the immune system and help you overcome colds.
Read Food, Drinks during Dengue: What to consume, what to avoid.
Additionally, you can add honey with your warm lemon water to treat a sore throat and cough. A small 2017 study found that honey helps to speed up the recovery of a sore throat as honey has anti-inflammatory and anti-bacterial properties.
Ginger tea
Traditionally, people use ginger tea to calm a sore throat. A 2013 study has shown that fresh ginger tea might help protect you from the respiratory syncytial virus — a highly infectious virus that commonly affects young children.
Besides, ginger is loaded with anti-inflammatory and antioxidant properties. If you drink ginger tea in the morning and before going to sleep, it soothes your sore throat.
Read Masala Tea Health Benefits: How Can It Improve Your Immunity
Moreover, ginger tea provides relief from motion sickness and alleviates nausea from morning sickness or chemotherapy. Ginger tea can also manage blood pressure and support heart health. It is also excellent to manage weight and blood sugar levels. The properties of ginger can fight against pain, inflammation, and cancer.
Chamomile tea
If you don’t like ginger tea, you can take chamomile tea, which is made from dried Matricaria flowers and is loaded with various healing properties. Research shows that chamomile tea can reduce inflammation in the throat.
Additionally, Chamomile tea is generally caffeine-free and so it is excellent to promote relaxation and help you sleep. Proper and adequate sleep boost your immune system and help you quick recovery from illness.
Read Health benefits of Chamomile Tea
Plus, chamomile tea has anti-inflammatory properties which minimize swelling and also repair tissues and reduce a cough.
Peppermint tea
You can also take peppermint tea which is another warm beverage to ease sore throat symptoms. Peppermint tea contains spearmint which has anti-inflammatory effects to reduce swelling and soothe a sore throat.
Besides, if you can inhale the steam from peppermint tea, it can treat other symptoms such as nasal congestion, and help you to breathe better.
Read 7 Yummy Coffee Recipes to Try at Home without Coffee Maker
Warm soup
You can take chicken noodle warm soup which is considered as is a classic home remedy for common illnesses.
Any kind of warm soup can be a great way to soothe your sore throat and get some key nutrients when you're feeling sick. Chicken broth has fatty acids and protein. Protein and fatty acids help your body build and repair healthy muscle, bone, skin, and blood cells.
Selenium that is available in chicken broth helps prevent and manage cardiovascular diseases and complications, including strokes, and high cholesterol.
Read Healthy Winter Soup Ideas and Recipes to Help You Get Through the Cold Weather
Moreover, chicken broth is enriched with vitamins and minerals, which are useful against common ailments like the common cold, the flu, and food poisoning.
Smoothie
When you're sick, you might experience a decreased appetite that can make it more difficult to get all the nutrients your body needs to fight off an illness.
So, you need a nutrient-packed smoothie as it is made with fruits and vegetables that are loaded with antioxidants — molecules to boost our immune system.
Read Child-friendly Winter Drinks to Keep Your Family Healthy, Warm, and Cozy
Some antioxidant-rich fruits and vegetables to make your smoothies: Blueberries, Strawberries, Mangoes, Kale, Spinach
Turmeric milk
Turmeric has powerful antioxidant properties to heal wounds. The properties of turmeric also fight off several infections like colds and sore throats. If you can drink hot turmeric tea or milk for a sore throat, you can cure your sore throat within 1-2 days.
Turmeric milk is anti-inflammatory also and contains the compound curcumin which boosts immunity. Turmeric also helps to reduce the risk of heart disease and improves digestion. It is also good for our skin and improves bone health.
Read 7 Healthy Warm Drinks to Stay Warm This Winter
Bottom Line
Sore throats are common and natural, especially during cold and flu season and winter season. While you are experiencing this condition, it is important to provide your body with the nutrients it needs to fight the infection. You can try homemade healing drinks to soothe your throat pain. So far, we have discussed 7 sore throat remedies to try at home. Hope it helps!
Food, Drinks during Dengue: What to consume, what to avoid
Every year a huge population is affected by Dengue fever. The main risk factor of dengue is the reduction of blood’s platelet level to a great extent that it could be fatal. The body requires sufficient vitamins, minerals, and protein to produce platelets. Patients diagnosed with dengue fever, need to consume the right diet for a fast recovery from dengue fever. Let’s take a look at the recommended foods and drinks during dengue fever.
Best Foods and Drinks for Dengue
Papaya leaves
Different studies have shown that the extract of the leaves of papaya is used as a complementary treatment for disorders of platelet function. Juice of Papaya leaves can help in increasing the platelet count in blood and boosts the immunity of your body.
Enzymes like papain and chymopapain are available in papaya extracts that prevent bloating and other digestive disorders. Papaya leaves juice is considered to be one of the best food items to increase platelets in the blood of patients suffering from dengue fever.
Read Dengue Fever: Symptoms, diagnosis, treatment, prevention
Vegetables
Dengue patients should eat vegetables like spinach, pumpkin, paprika, carrot, watercress, celery, broccoli, and beetroot to trigger the platelet levels in the blood. These vegetables help you purify your body. Make sure the vegetables are properly washed and cleaned before eating or cooking.
Spinach is enriched with protein and Vitamin K. These vitamins participate in the metabolism of coagulation factors and help to reduce the risk of excessive bleeding.
Take pumpkin with seeds, as they have amino acids and vitamins essential for platelet formation. If you regularly eat pumpkin, including seeds, it improves the absorption of proteins and vitamin A. These nutrients participate in the production of these small cells.
Read Why do Mosquitoes bite me so much?
Beetroot is rich in antioxidants that help prevent platelet reduction due to the effects of free radicals. It has vitamins A, C, and K and essential minerals, that help prevent the deterioration of blood vessels and the development of circulatory problems. By drinking beetroot juice regularly Dengue patients can strengthen their body's immunity.
Fruits
To increase the platelet count in the blood Dengue patients need both Vitamin C and Vitamin K. So while recovering from Dengue, the patients need to eat citrus fruits like lemon, orange, kiwi, or tangerines, and other fruits like strawberries, raspberries, tomatoes, or currants also.
Green Coconut water
Green coconut water is always best and it has huge health benefits. It is also a popular and effective drink to raise platelet levels. Coconut water bestows vitamins A, B, and C, and minerals, calcium, potassium, magnesium, iron, and phosphorus also.
Read Conjunctivitis: Causes, Symptoms, Treatment and Prevention
Besides, it is perfect for dengue patients as they experience dehydration as a result of high fever, diarrhea, anorexia, vomiting, and nausea during the febrile phase (days 4-6).
Nuts
As nuts and almonds are good sources of proteins and proteins are essential for normal platelet count, Dengue patients can take sufficient amounts of nuts during dengue fever.
Aloe Vera
Aloe Vera is full of nutrients including calcium, magnesium, phosphorus, potassium, zinc, sodium, copper, and vitamins C, E, and B.
Read 10 houseplants that are bad for your pets
Therefore, aloe vera juice has huge medicinal applications and it is an excellent ally in taking care of cardiovascular health and promoting platelet production in blood. Dengue patients can keep this food in their diet.
Yogurt
Yogurt is known for having probiotics-active bacteria. These active bacteria are very useful for keeping the intestines free of harmful germs and bacteria that are capable of causing diseases.
Dengue patients need to take Yogurt regularly to keep their defenses high. But not more than 150 grams of yogurt a day to get its healing benefits.
Read How to entertain an unwell child without screen time
Chicken Soup
Scientific evidence is available that chicken soup is great to stop the symptoms of colds and flu. Chicken soup is rich in Vitamin B12, Vitamin C, Antioxidants, Vitamin A, Vitamin D, Vitamin E, Vitamin K, and Carnosine.
These nutrients are essential to stimulating the defenses so that healing is carried out quickly. It will also help Dengue patients stay hydrated and raise the temperature of the airways, which is very effective in loosening mucus.
Green tea
Green tea is called one of the most popular healing drinks. It has a high content of polyphenols, very valuable anti-oxidant compounds with immunostimulatory effects.
Read Best foods for expecting mothers
A scientific study shows that a particular type of phenolic compound, called catechins, has the ability to eliminate some types of influenza viruses.
So, green tea should be included in the Dengue fever diet.
Neem Leaves
Neem leaves are blessed with chemicals such as Nimbin and Nimbidin that have anti-microbial, anti-inflammatory, and antipyretic properties. Research shows that these leaves have positive results in treating dengue by increasing blood platelets and white blood cell count.
Read How to Apply for Indian Medical Visa from Bangladesh
Foods and Drinks to Avoid while Recovering from Dengue Fever
Patients need to avoid certain foods that contain salicylates when infected with dengue fever. These types of food make blood thinner and slow clotting.
Non-vegetarian Foods
During the infection of dengue fever the human body becomes weak, the digestive system slows down, and non-vegetarian food does not easily digest. Therefore, while recovering from dengue fever, patients must strictly avoid non-vegetarian food.
Fried or Oily Foods
Dengue patients should also avoid oily foods burgers, pizza, potato chips, fried chicken, doughnuts, etc as these foods contain an increased amount of fat and can lead to high blood pressure and cholesterol.
Read How to Stay Physically Active during Pregnancy
Spicy Foods
Don’t take spicy foods at all during dengue fever as they can cause acid accumulation in the stomach and result in ulcers and damage to the wall. Eating these types of food during dengue infection may hinder the recovery process.
Caffeinated Beverages
Dengue fever causes dehydration and hence Dengue patients must drink plenty of fluids. Remember, caffeine, i.e. caffeinated beverages can revive the gastronomical tract, leading to nausea and diarrhea.
Besides, this type of beverage can increase the symptoms and production of stomach acid and cause rapid heart rate, and muscle breakdown which can hinder the dengue recovery process.
Read Should You See a Therapist? 8 Surefire Signs You Need Help
Bottom Line
Dengue is a curable disease but a little carelessness can cause serious issues and lead to death also. Hence, you need to be conscious about taking food and drinks. We have discussed here the foods and drinks to eat and avoid during dengue fever for quick recovery. Hope it helps!
Dengue Fever: Symptoms, diagnosis, treatment, prevention
The Aedes mosquito causes dengue fever. If you are bitten by a mosquito that carries the dengue virus, you will get dengue fever. The infection should not be neglected. If Dengue fever progresses to a severe stage, it can threaten life. This article will discuss the symptoms, treatments, home remedies, and preventions for dengue fever.
What is Dengue?
Dengue is a viral infection that is transmitted by the Aedes mosquito. The disease is found in tropical and subtropical regions of the world, and it is estimated that there are over 400 million cases of Dengue each year. The virus that causes Dengue is called DENV, and there are four DENV serotypes. It is possible to be infected four times with the virus.
However, there is no specific treatment for Dengue, but it can be prevented by protecting against mosquito bites. The Aedes mosquito typically lays its eggs in standing water, so removing sources of standing water can help to reduce the risk of dengue transmission.
Read Conjunctivitis: Causes, Symptoms, Treatment and Prevention
Signs of Dengue Fever
There are some symptoms to look for to know whether it is dengue fever or not. Such as feeling cold, loss of appetite, body aches, headache, pain in the eyes, lower back pain, muscle pain, joint pain, rash, fatigue, swollen lymph nodes, and skin-eye redness.
In the first few hours of dengue infection, pain is felt in the legs and joints. Fever can rise very quickly to 104 degrees Fahrenheit. Heart rate and blood pressure are low during this time. A red or pink rash may appear on the face, which then disappears.
High fever and other dengue symptoms last for 2-4 days, and then the body temperature drops rapidly with sweating. Body temperature remains normal for about a day before the second rapid rise in fever, and feels healthy. Later, an itchy rash appears all over the body except the face. The palms of the hands and soles of the feet may be bright red and swollen.
Read How to entertain an unwell child without screen time
Severe Symptoms of Dengue
Sometimes dengue infection is very serious or fatal. It is called severe Dengue. Symptoms of severe Dengue appear 3-7 days after the initial symptoms of Dengue. People who have been infected with Dengue before are at higher risk of severe Dengue.
The main symptoms of severe Dengue are severe abdominal pain, abdominal swelling, vomiting blood, bleeding from the gums, bleeding under the skin, difficulty or rapid breathing, feeling cold or sweating, rapid pulse, and drowsiness/loss of consciousness. If you see the symptoms of severe Dengue, you should go to the emergency department of the hospital without a moment's delay.
How to Diagnose Dengue Fever
If you see any dengue symptoms or have a serious fever, you should consult a registered doctor for further investigations. Not every fever is Dengue. Self-diagnosed for Dengue leaves other common causes of fever, such as pneumonia, urinary tract infection, typhoid, flu, etc., undiagnosed.
Read Best foods for expecting mothers
Patient's signs, symptoms, and physical examination data can help to diagnose the disease. However, results may vary from the first day to the next 10 days of dengue fever in different tests. So, self-testing can be confusing. Therefore, it would be wise to let the doctor understand first.
Doctors initially diagnose Dengue through NS1, CBC, IgG, and IgM. A maximum of Tk. 500 can be charged for the dengue NS1 Antigen test. A maximum of Tk. 400 can be taken for CBC (RBC + WBC + Hematocrit) test. Apart from this, IgG and IgM tests should be done within 500 Takas.
Treatment and Home Remedies of Dengue Fever
There is no specific treatment for dengue fever, but there are ways to ease the symptoms. Pain relievers, rest, and fluids can help to reduce fever and pain. In severe cases, hospitalization may be necessary in order to receive intravenous fluids and close monitoring.
Read Why do Mosquitoes bite me so much?
At the initial stage, take rest and drink plenty of water or liquid food. If the body is hydrated, headaches and muscle pain will be reduced. Drink coconut water, lemon juice, fruit juice, and saline at home.
Paracetamol can be taken to reduce the fever. However, a doctor's consultancy is needed if the patient has heart, liver, and kidney-related complications.
Aspirin, Clofenac, and Ibuprofen-like medicines should not be taken for body pain when suffering from dengue fever. Taking such medicines during Dengue can cause bleeding.
Read How to Apply for Indian Medical Visa from Bangladesh
What not to do
Platelets are no longer a key factor in dengue fever. There is no need to worry about the platelet count. If the platelet count falls below 10,000 or if there is bleeding from any part of the body, platelets or fresh blood can be given if necessary. However, such situations are very rare.
Many people say that eating papaya leaf juice etc. increases platelets. Actually, these have no role. After the fever subsides, platelets begin to grow by themselves after the crisis period has passed. At the end of the fever, the blood pressure may drop, or there may be bleeding from the gums, nose, or rectum. If this is the case, intravenous saline may be needed. In these cases, hospital admission is required.
When to go to the hospital
In the case of Dengue, what kind of treatment to take, staying at home or in a hospital, depends on its type or category. There are three types or categories of dengue fever - 'A,' 'B,' and 'C.' Patients in the first category are normal. They only have a fever. Most dengue patients are in the 'A' category. They do not require hospitalization. Resting at home is enough.
Read Top 10 Bathroom Plants to grow in humid environment of shower
Category 'B' dengue patients may require hospitalization. Certain symptoms, such as abdominal pain, vomiting, diabetes, obesity, pregnancy, congenital problems, and kidney or liver problems, are recommended for hospitalization.
Category 'C' dengue fever is the worst. It can damage the liver, kidneys, and brain. In some cases, an intensive care unit or ICU may be required.
So, you have to follow the doctor's instructions.
Read Motorcycle Accident Prevention: Tips to Reduce the Risk of a Crash
Dengue Fever Season in Bangladesh
Generally, the dengue outbreak occurs from July to October in Bangladesh, as the Aedes mosquito spreads during this period. But we have to be careful throughout the year as in some years; Dengue started in June.
Preventive Measures for Dengue Fever
The Aedes mosquito usually lays its eggs in clear water. So, make sure water does not stay for more than three to five days. This water can accumulate anywhere. Aedes mosquitoes breed in the water collected on the roof of the house or in the flower tub of the balcony, at various points of the building under construction, tires lying on the side of the road, or in other containers.
So, check if there is any stagnant water in any places around the house. Also, use mosquito nets and spray in the house. Additionally, use mosquito repellent cream while going out or staying at home.
Read Should You See a Therapist? 8 Surefire Signs You Need Help
Final Words
In Bangladesh, the Dengue death toll is rising every year. Especially in the cities and towns, many inhabitants are being affected by Dengue fever. Some prevention measures for Dengue can minimize the spread of the disease.
However, all fevers are not Dengue. So, make sure you know the sign and symptoms of Dengue Fever to determine if you have it or not. You may also get affected by viral fever too. So far, we have discussed the diagnosis process of Dengue fever. Proper consultations with doctors are essential to avoid serious medical conditions. If proper treatment for Dengue fever is provided the patient can be cured.
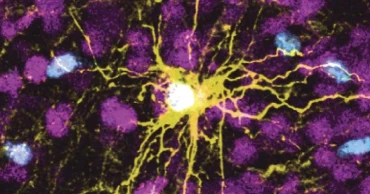
Scientists grow human brain tissue in rats to study diseases
Scientists have transplanted human brain cells into the brains of baby rats, where the cells grew and formed connections.
It’s part of an effort to better study human brain development and diseases affecting this most complex of organs, which makes us who we are but has long been shrouded in mystery.
“Many disorders such as autism and schizophrenia are likely uniquely human” but “the human brain certainly has not been very accessible,” said said Dr. Sergiu Pasca, senior author of a study describing the work, published Wednesday in the journal Nature.
Approaches that don’t involve taking tissue out of the human brain are “promising avenues in trying to tackle these conditions.”
The research builds upon the team’s previous work creating brain “organoids,” tiny structures resembling human organs that have also been made to represent others such as livers, kidneys, prostates, or key parts of them.
To make the brain organoids, Stanford University scientists transformed human skin cells into stem cells and then coaxed them to become several types of brain cells. Those cells then multiplied to form organoids resembling the cerebral cortex, the human brain’s outermost layer, which plays a key role in things like memory, thinking, learning, reasoning and emotions.
Scientists transplanted those organoids into rat pups 2 to 3 days old, a stage when brain connections are still forming. The organoids grew so that they eventually occupied a third of the hemisphere of the rat’s brain where they were implanted. Neurons from the organoids formed working connections with circuits in the brain.
Human neurons have been transplanted in rodents before, but generally in adult animals, usually mice. Pasca, a psychiatry professor at the Stanford School of Medicine, said this is the first time these organoids have been placed into early rat brains, creating “the most advanced human brain circuitry ever built from human skin cells and a demonstration that implanted human neurons can influence an animal’s behavior.”
To examine a practical use of this approach, scientists transplanted organoids into both sides of a rat’s brain: one generated from a healthy person’s cells and another from the cells of a person with Timothy syndrome, a rare genetic condition associated with heart problems and autism spectrum disorder.
Five to six months later, they saw effects of the disease related to the activity of the neurons. There were differences in the two sides’ electrical activity, and the neurons from the person with Timothy syndrome were much smaller and didn’t sprout as many extensions that pick up input from nearby neurons.
Researchers, whose study was funded partly by the National Institutes of Health, said they could do the same sorts of experiments using organoids made from the cells of people with disorders such as autism or schizophrenia — and potentially learn new things about how these conditions affect the brain, too.
Dr. Flora Vaccarino of Yale University – who previously grew lumps containing cerebral cortex that were made with DNA from people with autism – said the study moves the field forward.
“It’s extremely impressive what they do here in terms of what these cells can actually show us in terms of their advanced development … in the rat,” said Vaccarino, who wasn’t involved with the study.
Such experiments in animals raise ethical concerns. For example, Pasca said he and his team are cognizant of the rats’ well-being and whether they still behave normally with the organoids inside them, which he says they do. Still, Pasca does not believe this should be tried in primates. Ethicists also wonder about the possibility of brain organoids in the future attaining something like human consciousness, which experts say is extremely unlikely now.
Some scientists are studying human brain organoids outside of animals. For example, researchers at ETH Zurich in Switzerland published a study in Nature earlier this month describing how they are growing brain-like tissue from stem cells in the lab and then mapping the cell types in various brain regions and genes regulating their development. Some are using these structures to study autism.
Pasca said brain organoids could also be used to test new treatments for neuropsychiatric disorders, the largest cause of disability worldwide. Such research, he said, should help scientists make strides that have been extremely difficult until now because it’s so hard to get at the human brain – which is “the reason why we’re so much more behind in psychiatry compared to any other branch of medicine in terms of therapeutics.”
Conjunctivitis: Causes, Symptoms, Treatment and Prevention
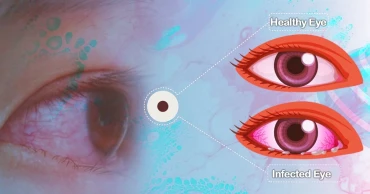
The medical name of Pink Eye is Conjunctivitis. It is one of the most common eye infections in children and adults. Viruses, bacteria, or allergens are responsible for pink eye. If you or any of your family members experience such conditions, try to find out whether the pink eye is caused by allergies or infection; as each condition has different treatments. Let’s take a look at the causes, symptoms, treatments, and preventions of Conjunctivitis.
Symptoms of Conjunctivitis
Actually, pink eye is an inflammation of the conjunctiva that lines a human's eyelid and covers the white part of your eyeball. With pink eye, the white part becomes light pink to reddish.
Common signs of Conjunctivitis are as follows:
-Becomes red or pink in the white part of the eye
-Increased amount of tears
-Itchy eyes and nose
-Blurred vision
-Sneezing
-Swelling of the eyelid
Read Why do Mosquitoes bite me so much?
When you are affected by Conjunctivitis, you may experience these symptoms usually in both eyes but not always equally.
If you have any of these persistent symptoms, make sure you consult an ophthalmologist.
Causes of Conjunctivitis
-The main cause of the allergy to pink eye or conjunctivitis is the body's reaction to allergens including pollen from trees, plants, grasses, and weeds; dust mites; molds; dander from pets; medicines; or cosmetics.
-It happens often among people with other allergic conditions like hay fever, asthma, and eczema.
Read How to Control Your Meat Addiction?
Home remedies for Pink Eye or Conjunctivitis:
Check here 6 effective home remedies that can help you treat allergic conjunctivitis:
Cold compress
A cold compress is a comfort measure you can take that can provide you instant relief. To apply this remedy take one or more small pieces of linen cloth and then dip the cloth in cold water. It would be better if you dip the cloth in ice-cold water. Remember, you have to remove the excess water from the cloth before applying it to your eyes.
This cold compress is so useful that it can help you reduce itchiness and swelling caused due to an unknown substance that has entered the eyes. You need to repeat this system several times for at least 5-10 minutes. To get good results it is recommended to use different cloth pieces for both eyes to avoid the infection from increasing.
Read How to Apply for Indian Medical Visa from Bangladesh.
Saline solution
Another effective home remedy against allergic pink eye or conjunctivitis is washing your eyes with saline solution. A saline solution is a concoction of common salt and water. So, you can use the saline solution as an eyewash as the saline solution helps decrease inflammation and irritation in the eyes. However, never use hot saline solution which can cause more harm to your eyes than good.
Rose water
Naturally, rose water is excellent as it has anti-inflammatory and soothing resources. Rose water is not only cool but soothing for the eyes also. It is great and helps to get rid of the dust particle that caused allergic conjunctivitis in the first place.
If you are suffering from allergic conjunctivitis, you can use a few drops of rose water a day in the infected eye. Uses of 2-3 days can help you treat allergic conjunctivitis. Besides, you can use cotton balls dipped in rose water over your eyes to minimize the irritation of your eyes.
Read Top 10 Bathroom Plants to grow in the humid environment of the shower
Aloe vera gel
Aloe vera has compounds namely aloin and emodin that have anti-bacterial and anti-viral effects. So, aloe vera gel is an important and effective home remedy for allergic conjunctivitis or pink eye.
Moreover, aloe vera helps to bring down inflammation and speeds up the healing process against the allergy caused due to dust particles. You can apply aloe vera gel around the infected area as soon as you notice the symptoms of allergic pink eye. Generally, aloe vera does not have any inauspicious effects on the cornea of the eye.
Turmeric
Usually, turmeric powder is famous for its many known healing properties. Besides, it is very effective and result-oriented against allergic conjunctivitis or pink eye. You can use turmeric to soothe your affected eye because it helps to reduce inflammation of the eyes. Because of its antibacterial properties, it can prevent any further infection of the eye.
Read Motorcycle Accident Prevention: Tips to Reduce the Risk of a Crash
Take a piece of cotton or cloth and soak it in a pot of boiled turmeric water. Then use the soaked and warm cloth piece to treat allergic conjunctivitis. It will also work as a warm compress. But don’t use so much hot cloth.
Chamomile tea bag
Chamomile tea is another excellent cold compress or an eyewash. When you are suffering from swelling and itchiness in the eye, you can use this remedy. Take two tea bags of chamomile tea and dip them for 5 minutes in a cup of hot water.
When they become cool, put them in the refrigerator and apply them as a cold compress over the eyes. For better results, do this 3-4 times a day for 2-3 days. As chamomile tea has anti-microbial properties, it is an effective home remedy for allergic conjunctivitis.
Read Heat Stroke Prevention: Best foods, drinks to avoid heat exhaustion
Preventive measures for Conjunctivitis
-Don't touch your eyes with your hands unnecessarily.
-Wash your hands frequently.
-Use a clean towel and washcloth always.
-Don't share towels or washcloths with others.
-Change your pillowcases when you feel they are not clean.
-Don’t use eye cosmetics like mascara.
-Don't share eye cosmetics or personal eye care items with other people.
Read 10 Healthy Bedtime Teas for Better Sleep, Relaxation
Bottom Line
Pink eye or Conjunctivitis can result from many causes. However, it is not highly contagious like infected pink eye. Home remedies are the most common and effective treatments for allergic pink eye, and in most cases, you can relief from it without prescription drugs. To prevent transmission, make sure you practice good hand washing and avoid touching the eyes and face every now and then. Of course, go to your doctor if you feel something serious.